Edit this Installation Qualification Template into Your Protocol
Installation Qualification Template Rationale.
First step is to ensure your Validation Installation Qualification template is 21 CFR Part 210/211/820/11 compliant and has an in-built “follow-me” completion SOP. Completion is then trouble free
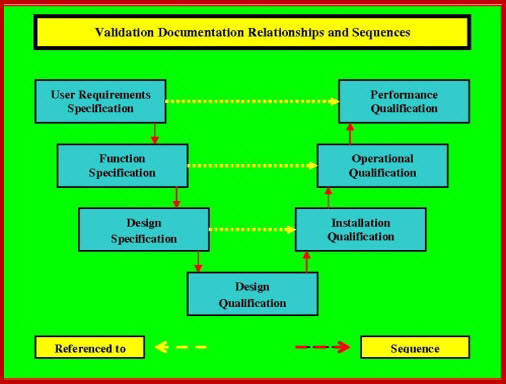
The Installation Qualification Template execution; verifies that the equipment, and its ancillary systems or sub-systems have been installed in accordance with installation qualification template drawings and or specifications. It further details a list of all the cGMP requirements that are applicable to this particular installation qualification template. These requirements must all be satisfied before the IQ can be completed and the qualification process is allowed to progress to the execution of the Operational Qualification (OQ).
In the past equipment suppliers have often been delinquent in supplying the right scope and quality of support documentation for their products. As a consequence end user companies have not always had sufficient information (especially in the form of engineering drawings and specifications) about the equipment they have purchased, to develop and put into place the quality of documentation that regulatory requirements demand. For correct and trouble free qualification it is really essential that the scope and quality of the proposed equipment documentation; is specified at the equipment procurement stage.
Installation Qualification Template Introduction
Our Installation Qualification Template (IQ) protocol comes with an interactive SOP as an attached prefixed document. As you follow the installation qualification template protocol requirements as specified in the SOP, you complete the actual IQ protocol. This makes it a really easy and straight forward document to use. The Installation Qualification template is normally a stand alone document, however, with careful pre-planning, certain aspects of the IQ activities can be integrated with the Factory Acceptance Testing (FAT), and the equipment Commission Testing.
Conformance with current Good Manufacturing Procedures. (cGMP's) requires, that what ever approach is used, it is fully documented in the individual Validation (Master) Plan (VMP or VP). The IQ should not start with the Factory Acceptance Testing (FAT) or Commissioning tasks, but it should start before these tasks are completed; enabling the validation team to witness and document the final FAT and commissioning testing. The integration of these activities greatly reduces the costly and time consuming replication of unnecessary retesting.
There is a grey area of testing / inspection in the transition from the Installation Qualification Template (IQ) that is open to rationalization, i.e. it has to be done, but it can satisfactorily be included in either the IQ or OQ. On most projects the simpler the IQ is kept, the quicker it is completed, reviewed and out of the way, allowing progress to the OQ. It can therefore pay dividends to keep the IQ as basic as possible. Where possible, the Installation Qualification Template can be kept to the pre-powering up stage. Keeping the IQ at this level allows healthy project progress. There should be no powering up or utility problems to hinder IQ sign off, and rapid progress through the OQ to the Performance Qualification (P1Q) and where applicable the Process Qualification (P2Q), becomes feasible. It can be reasoned that as soon as you power up, you enter the equipment operation stage, where verification of the User Requirements Specification (URS) and 21 CFR Part 11 verification testing are undertaken.
For this reason we have modulized the Installation qualification template (IQ) and OQ, building in the tests and inspections that are not in this grey area, and building the one's that are, as modules that can be added to either the IQ or the OQ. This has not been done just because it might work, this has been advocated by us for some time. We have experience major delays to projects, caused purely by waiting for the client to complete document reviews and approvals. See Comment 9 in cGMP-FDA-483.
Installation Qualification Template Scope (IQ).
Learn how to edit the Installation Qualification template into a first class Installation Qualification Protocol. The Installation Qualification Template scope and intensity of this IQ will cover the under listed subjects, but is not restricted to them alone.
- Verification that all components parts ~.
- Verify installation is as specified ~.
- Insert a brief description of what part of the validated product process ~~..
- Insert a brief description of the operational function ~~. .
- Verify supply voltages conform with ~~.
- Verify that electrical installation qualification template complies with ~~.
- Verify that all alarms and visual displays are ~~.
- Verify that where used, the correct versions of ~~.
- Confirm that the issue level of software ~~.
- Identify and verify that serial numbers and model number of all ~~.
- Review all calibration certificates for ~~.
- Verify where used, there is signals continuity~~.
- Verify in installation qualification template- signal continuity ~~.
- Verify that the ambient conditions are ~~.
- Verify that the documentation provided by the ~~ maintenance of the system ~~.