Click anywhere on this image for further details.
Click anywhere on this image for further details.Understanding how to write a superb Facilities HVAC qualification protocol requires a diverse skills set covering controls and management of environmental parameters, such as temperature, humidity and particle content along with biological contamination. All are critical aspects of the mandatory cGMP tests and inspections ing facilities HVAC system must be performed.
This building Habitat validation must utilize a risk-based approach, which involves a high level of process understanding and focuses on potential problems, risks, and all suitable countermeasures. The facilities HVAC validation must verify that in all areas where drugs or raw materials have air contact, the air cannot adversely affect the quality of the product.
To fulfill regulatory requirements, a clean room facilities HVAC system must maintain environmental condition within specified specifications. These conditions must be regularly monitored to ensure constant awareness of environmental conditions, such as temperature, humidity, pressure and particle levels. Digital outputs for alarms must also be monitored to ensure that an alarm will be raised if there is a change in any of the designated parameters.
All such environmentally controlled areas, zones and or rooms must be included in a preventative maintenance and calibration program that will monitor all trends and ensure continued use within the User Requirements specified specifications.
Developing Continuous Process monitoring.
Statistical Process
Control (SPC).
PC systems use statistics to monitor the quality of products
created by a process and send alerts when something abnormal is detected. Control charts are pretty good at showing
when process qualification is behaving normally and are usually the first step in
controlling quality.
R2R control involves the use of measurement data to optimize
a particular process step or set of steps
This is an example a feedback control system. R2R control systems generally start off
controlling a single process qualification task, but can be grown to achieve factory-wide control
solutions.
Fault Detection and
Classification (FDC).
FDC systems monitor the performance of manufacturing
equipment during operation and detect events that might affect the quality of
the product. When an issue is detected a
warning message is raised.
Advanced Process
Control (APC).
APC brings together all of these pieces of software. In a fully realized APC system, each
component (SPC, R2R, FDC) can interact with the others to form a comprehensive
factory control system.
Introduction to Facilities HVAC.
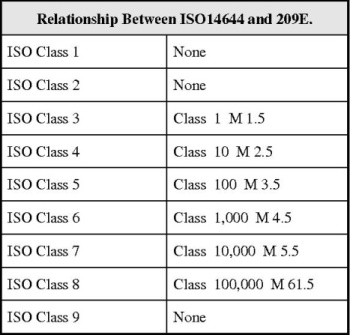
Where Facilities HVAC environmental parameters, such as temperature, humidity, particle content and biological contamination are important aspects of pharmaceutical, biotechnical and or medical device production, validation and qualification of the facilities HVAC system must be performed.
This building Habitat validation must utilize a risk-based approach, which involves a high level of process understanding and focuses on potential problems, risks, and all suitable countermeasures. The facilities HVAC validation must verify that in all areas where drugs or raw materials have air contact, the air cannot adversely affect the quality of the product.
To fulfill regulatory requirements, a clean room facilities HVAC system must maintain environmental condition within specified specifications. These conditions must be regularly monitored to ensure constant awareness of environmental conditions, such as temperature, humidity, pressure and particle levels. Digital outputs for alarms must also be monitored to ensure that an alarm will be raised if there is a change in any of the designated parameters.
All such environmentally controlled areas, zones and or rooms must be included in a preventative maintenance and calibration program that will monitor all trends and ensure continued use within the User Requirements specified specifications.
Energy Requirements and Efficiencies.
A fundamental problem is that in conventional facilities HVAC systems; with conventional cooling coils (whether using chilled water or direct-expansion refrigerant) the latent loads from ventilation on very humid days can be excessive. All conventional chillers and air conditioners are essentially sensible cooling devices that dehumidify by lowering air temperature below its dew-point so that moisture condenses. These systems must run with a wet cooling coil and the air that leaves this coil must be close to saturation.
The limitation of conventional chillers and direct-expansion (DX) air conditioners becomes evident when one tries to use them in an advanced facilities HVAC system. Technologies such as displacement ventilation, chilled beams, and radiate panels can be part of a low-energy facilities HVAC system that eliminates the fan energy used in a conventional system that recirculates large volumes of air. However, these advanced systems will not work with a conventional chiller or DX air conditioner that supplies relatively cold air (e.g., 50 to 55 F) that is saturated with moisture (i.e., 100% rh). What is required is a cooling system that supplies drier, but warmer air.
Typical supply air conditions for displacement ventilation are 65 F and 50% relative humidity. This supply air has an absolute humidity of 45.5 grains moisture per pound of dry air (which is equivalent to a humidity ratio of 65 g/kg and a dewpoint of 46.0 F). Any cooling system that dehumidifies air by reducing its temperature to condense the moisture must first cool air to below 46 F and then reheat the air to 65 F. As shown on the neighboring psychometric chart, a cooling system that process 6,000 cfm of warm, humid outdoor air must do 54.0 tons of cooling and then 10.8 tons of reheating. This air conditioner is doing 25% more cooling than is required to meet the load. Furthermore, this percent excess cooling becomes much larger during cooler, damp weather, e.g., if it were 70 F and raining, over-cooling would be 42% of the required cooling.
This cycle is seldom understood by management, who consequently fail to realise that the rise in energy consumption of a system 'out of calibration' can be very substantial. These systems should be subjected to annual calibration and a defined method of tuning to attain minimum energy consumption.
Documents Available.
This combined facilities HVAC SOP and IQ template makes it so easy for you to raise a quality IQ. With the new suite of facilities HVAC documents from the DQ - IQ - OQ - PQ, all user friendly and ready to go, your facilities HVAC validation has got much simpler. The final product is a professional and comprehensive Heating Ventilation and Air Conditioning Installation Validation Protocol. One that you can produce in less than 60 minutes. Yes, think about it, we all know how long producing IQ documents has taken in the past. There is now no reason for not being able to produce 4 to 8, IQ protocols per 8 hour day, or a complete suite in one day.
PLEASE CLICK HERE TO GO TO SHOP AND PURCHASE THIS DOCUMENT
Facilities HVAC Performance Validation.
This fully formatted Facilities HVAC Performance Validation is the last in the chain of qualifying tests that facilities HVAC systems are subjected to, before being considered qualified. However environmental validation is required to verify air quality. This easy to use Facilities HVAC Validation SOP and Protocol, can be simply and quickly converted (using find replace techniques) into your own company bespoke document. Now you can purchase these document in a suite DQ to PQ, getting you ready at express speed to qualify a system. It has never been quicker or simpler.
21 Years of retailing cGMP - 35k templates supplied to the industry.
Validation Plan (Issue 8)
$89.00 10000008
This document follows our well developed method of using a generic document and allowing the customer to apply an attached detailed SOP to it, turning the generic document quickly into a first class company bespoke document. This VP details and integrates all validation activities and procedures required for a small to medium sized project, involving production/facilities/utility equipment/ facilities HVAC using electronic controls or monitoring.
Validation Risk Assessment (Issue-11.) $125.00
10000014
The Risk and Part 11 Validation Risk Assessment(VRA) protocol is becoming the most important document in the validation train. The VRA reassures the regulators that you have looked at specific equipment functionality and considered the appropriate level of validation that is required. You have also considered various aspects of its use and the implications of any malfunctions. From the results of this exercise the scope of all validation activity can and must be justified. This is a robust and simple to execute document, one that will lead you through the process and deliver a result that can be used as the foundation for your validation activities.
This VRA now includes the assessment table for categorizing and documenting the new 21 CFR Part 11 guidance ruling on what predicate data must be stored in a Part compliant system, along with the new broadsheet to establish your new database of part 11 records. (now mandatory). Applies to all facilities HVAC protocols.
Top of Form
Cold Chain Zone IQ (Issue 5)
$115.00 10000401
The SOP used to generate this IQ, takes you through the process line by line, chapter by chapter. It really is unique to find a SOP document so easy to use, all the work is done for you. All the documents are detailed, all the drawings listed and all the checks and tests detailed. The final product is a professional and comprehensive Installation Qualification Protocol for your temperature controlled cabinet or room. One that you can produce in less than 60 minutes. Yes, think about it, we all know how long producing IQ documents has taken in the past. This applies to all facilities HVAC protocols.